Rearranging Arrhenius Equation To Find Activation Energy
Determining the activation energy. The Arrhenius equation. Can be written in a non-exponential form which is often more convenient to use and to interpret graphically. Taking the logarithms of both sides and separating the exponential and pre-exponential terms yields. The value of the slope (m) is equal to -Ea/R where R is a constant equal to 8.314 J/mol-K. The activation energy can also be found algebraically by substituting two rate constants (k1, k2) and the two corresponding reaction temperatures (T1, T2) into the Arrhenius Equation (2). Have an energy greater than or equal to E a.! Temperature Dependence of the Rate Constant:! Increasing the temperature of a reaction generally speeds up the process (increases the rate) because the rate constant increases according to the Arrhenius Equation.! Rate (M s-1) = k AxBy! K=Ae −E a RT As T increases, the value of the exponential part of the.
All molecules possess a certain minimum amount of energy. The energy can be in the form of kinetic energy or potential energy. When molecules collide, the kinetic energy of the molecules can be used to stretch, bend, and ultimately break bonds, leading to chemical reactions. If molecules move too slowly with little kinetic energy, or collide with improper orientation, they do not react and simply bounce off each other. However, if the molecules are moving fast enough with a proper collision orientation, such that the kinetic energy upon collision is greater than the minimum energy barrier, then a reaction occurs. The minimum energy requirement that must be met for a chemical reaction to occur is called the activation energy, (E_a).
Figure 1: In Greek mythology Sisyphus was punished by being forced roll an immense boulder up a hill, only to watch it roll back down, and to repeat this action forever. If this were a chemical reaction, then it would never be observed, since the reactants must overcome the energy barrier to get to the other side (products).
The reaction pathway is similar to what happens in Figure 1. To get to the other end of the road, an object must roll with enough speed to completely roll over the hill of a certain height. The faster the object moves, the more kinetic energy it has. If the object moves too slowly, it does not have enough kinetic energy necessary to overcome the barrier; as a result, it eventually rolls back down. In the same way, there is a minimum amount of energy needed in order for molecules to break existing bonds during a chemical reaction. If the kinetic energy of the molecules upon collision is greater than this minimum energy, then bond breaking and forming occur, forming a new product (provided that the molecules collide with the proper orientation).
Figure 2: Reaction coordinate diagram for the bimolecular nucleophilic substitution ((S_N2)) reaction between bromomethane and the hydroxide anion. Image used with permission from Wikipedia.
The activation energy ((E_a)), labeled (Delta{G^{ddagger}}) in Figure 2, is the energy difference between the reactants and the activated complex, also known as transition state. In a chemical reaction, the transition state is defined as the highest-energy state of the system. If the molecules in the reactants collide with enough kinetic energy and this energy is higher than the transition state energy, then the reaction occurs and products form. In other words, the higher the activation energy, the harder it is for a reaction to occur and vice versa.
Effects of Enzymes on Activation Energy
However, if a catalyst is added to the reaction, the activation energy is lowered because a lower-energy transition state is formed, as shown in Figure 3. Enzymes can be thought of as biological catalysts that lower activation energy. Enzymes are proteins or RNA molecules that provide alternate reaction pathways with lower activation energies than the original pathways. Enzymes affect the rate of the reaction in both the forward and reverse directions; the reaction proceeds faster because less energy is required for molecules to react when they collide. Thus, the rate constant (k) increases.
Figure 3: Lowering the Activation Energy of a Reaction by a Catalyst. This graph compares potential energy diagrams for a single-step reaction in the presence and absence of a catalyst. The only effect of the catalyst is to lower the activation energy of the reaction. The catalyst does not affect the energy of the reactants or products (and thus does not affect ΔG).
As indicated by Figure 3 above, a catalyst helps lower the activation energy barrier, increasing the reaction rate. In the case of a biological reaction, when an enzyme (a form of catalyst) binds to a substrate, the activation energy necessary to overcome the barrier is lowered, increasing the rate of the reaction for both the forward and reverse reaction. See below for the effects of an enzyme on activation energy.
Catalysts do not just reduce the energy barrier, but induced a completely different reaction pathways typically with multiple energy barriers that must be overcome. For example:
Activation Enthalpy, Entropy and Gibbs Energy
In thermodynamics, the change in Gibbs free energy, ΔG, is defined as:
- ΔG = change in Gibbs free energy of the reaction
- ΔH = change in enthalpy
- ΔS = change in entropy
( Delta G^o ) is the change in Gibbs energy when the reaction happens at Standard State (1 atm, 298 K, pH 7). To calculate a reaction's change in Gibbs free energy that did not happen in standard state, the Gibbs free energy equation can be written as:

[ Delta G = Delta G^o + RT ln K label{2}]
where
- ΔG is change in Gibbs free energy of the reaction
- ΔGo is the standard Gibbs free energy
- R is the Ideal Gas constant (8.314 J/mol K)
- (K) is the equilibrium constant
When the reaction is at equilibrium, ( Delta G = 0). The equation above becomes:
[ 0 = Delta G^o + RTln K ]
Solve for ΔGo:
[ Delta G^o = -RT ln K ]
Similarly, in transition state theory, the Gibbs energy of activation, ( Delta G ^{ddagger} ), is defined by:
[ Delta G ^{ddagger} = -RT ln K^{ddagger} label{3}]
and
[ Delta G ^{ddagger} = Delta H^{ddagger} - TDelta S^{ddagger}label{4} ]
where
- ( Delta G^{ddagger} ) is the Gibbs energy of activation
- ( Delta H^{ddagger} ) is the enthalpy of activation
- ( Delta S^{ddagger} ) is the entropy of activation
[ ln K^{ddagger} = -dfrac{Delta H^{ddagger}}{RT} + dfrac{Delta S^{ddagger}}{R} ]
As shown in the figure above, activation enthalpy, (Delta{H}^{ddagger} ), represents the difference in energy between the ground state and the transition state in a chemical reaction. The higher the activation enthalpy, the more energy is required for the products to form. Note that this activation enthalpy quantity, ( Delta{H}^{ddagger} ), is analogous to the activation energy quantity, Ea, when comparing the Arrhenius equation (described below) with the Eyring equation:
[E_a = Delta{H}^{ddagger} + RT]
In general, a reaction proceeds faster if Ea and (Delta{H}^{ddagger} ) are small. Conversely, if Ea and ( Delta{H}^{ddagger} ) are large, the reaction rate is slower.
Calculation of Ea using Arrhenius Equation
As temperature increases, gas molecule velocity also increases (according to the kinetic theory of gas). This is also true for liquid and solid substances. The (translational) kinetic energy of a molecule is proportional to the velocity of the molecules (KE = 1/2 mv2). Therefore, when temperature increases, KE also increases; as temperature increases, more molecules have higher KE, and thus the fraction of molecules that have high enough KE to overcome the energy barrier also increases.
The fraction of molecules with energy equal to or greater than Ea is given by the exponential term (e^{frac{-E_a}{RT}}) in the Arrhenius equation:
[k = Ae^{frac{-E_a}{RT}} label{5}]
- k is the rate constant
- Ea is the activation energy
- R is the gas constant
- T is temperature in Kelvin
- A is frequency factor constant or also known as pre-exponential factor or Arrhenius factor. It indicates the rate of collision and the fraction of collisions with the proper orientation for the reaction to occur.
Taking the natural log of both sides of Equation (ref{5}) yields the following:
[ln k = ln A - frac{E_a}{RT} label{6}]
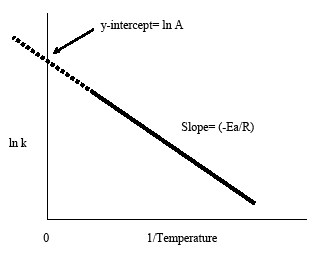
Equation (ref{4}) has the linear form y = mx + b. Graphing ln k vs 1/T yields a straight line with a slope of -Ea/R and a y-intercept of ln A., as shown in Figure 4.
Figure 4
Figure 5
As indicated in Figure 5, the reaction with a higher Ea has a steeper slope; the reaction rate is thus very sensitive to temperature change. In contrast, the reaction with a lower Ea is less sensitive to a temperature change. Because radicals are extremely reactive, Ea for a radical reaction is 0; an arrhenius plot of a radical reaction has no slope and is independent of temperature.
The activation energy can also be calculated directly given two known temperatures and a rate constant at each temperature. Using Equation (2), suppose that at two different temperatures T1 and T2, reaction rate constants k1 and k2:
[ln; k_1 = - frac{E_a}{RT_1} + ln A label{7} ]
and
[ln; k_2 = - frac{E_a}{RT_2} + ln A label{8} ]
Subtract (ln; k_2) from (ln; k_1):
[ ln; k_1 - ln; k_2 = left (- dfrac{E_a}{RT_1} + ln A right ) - left(- dfrac{E_a}{RT_2} + ln A right) label{9} ]
After rearrangement:
[ ln left (dfrac{k_1}{k_2} right ) = left(dfrac{1}{T_2} - dfrac{1}{T_1}right)dfrac{E_a}{R} label{10} ]
Questions
- Given that the rate constant is 11 M-1s-1 at 345 K and the pre-exponential factor is 20 M-1s-1, calculate the activation energy.
- If a reaction's rate constant at 298K is 33 M-1s-1and 45 M-1s-1at 675 K, what is the activation energy?
- What is the Gibbs free energy change at the transition state when ΔH at the transition state is 34 kJ/mol and ΔS at transition state is 66 J/mol at 334K?
- What is the Activation Energy of a reverse reaction at 679K if the forward reaction has a rate constant of 50M-1s-1, a pre-exponential factor of 30M-1s-1, and a ΔH of reaction of 23 kJ/mol?
- Enzymes lower activation energy, and thus increase the rate constant and the speed of the reaction. However, increasing the temperature can also increase the rate of the reaction. Does that mean that at extremely high temperature, enzymes can operate at extreme speed?
Solutions
1. Use the Arrhenius Equation: (k = Ae^{-E_a/RT})
- k is the rate constant, A is the pre-exponential factor, T is temperature and R is gas constant (8.314 J/molK)
- ln(11) = (20)e-Ea/(8.314)(345)
- Ea = 6084.1 J/mol
2. Use the equation: ( ln left (dfrac{k_1}{k_2} right ) = dfrac{-E_a}{R} left(dfrac{1}{T_1} - dfrac{1}{T_2}right))
- ln(33/45) = [-Ea/8.314](1/298 - 1/675)
- Ea = 1375.8 J/mol
3. Use the equation (Delta{G} = Delta{H} - T Delta{S})
- (Delta{G} = (34 times 1000) - (334)(66))
- (Delta{G} = 11956, J/mol)
4. Use the equation (ln k = ln A - dfrac{E_a}{RT})to calculate the activation energy of the forward reaction
- ln(50) = (30)e-Ea/(8.314)(679)
- Ea = 11500 J/mol
- Because the reverse reaction's activation energy is the activation energy of the forward reaction plus ΔH of the reaction:
- 11500 J/mol + (23 kJ/mol X 1000) = 34500 J/mol
5. No. Most enzymes denature at high temperatures. At some point, the rate of the reaction and rate constant will decrease significantly and eventually drop to zero. Once the enzyme is denatured, the alternate pathway is lost, and the original pathway will take more time to complete.
References
- Atkins P., de Paua J.. Physical Chemistry for the Life Sciences. pg 256-259. New York. Oxford Univeristy Press. 2006.
- Garrett R., Grisham C. Biochemistry. 3rd Edition. pg 64. California. Thomson Learning, Inc. 2005
- Wade L.G. Organic Chemistry. 6th Edition. pg 139-142. New Jersey. Pearson Prentice Hall. 2006.
Contributors
- Matthew Bui, Kan, Chin Fung Kelvin, Sinh Le, Eva Tan
In physical chemistry, the Arrhenius equation is a formula for the temperature dependence of reaction rates. The equation was proposed by Svante Arrhenius in 1889, based on the work of Dutch chemist Jacobus Henricus van 't Hoff who had noted in 1884 that van 't Hoff equation for the temperature dependence of equilibrium constants suggests such a formula for the rates of both forward and reverse reactions. This equation has a vast and important application in determining rate of chemical reactions and for calculation of energy of activation. Arrhenius provided a physical justification and interpretation for the formula.[1][2][3] Currently, it is best seen as an empirical relationship.[4]:188 It can be used to model the temperature variation of diffusion coefficients, population of crystal vacancies, creep rates, and many other thermally-induced processes/reactions. The Eyring equation, developed in 1935, also expresses the relationship between rate and energy.
- 4Theoretical interpretation of the equation
Equation[edit]
Arrhenius equation gives the dependence of the rate constant of a chemical reaction on the absolute temperature, a pre-exponential factor and other constants of the reaction.
where
- k is the rate constant,
- T is the absolute temperature (in Kelvin),
- A is the pre-exponential factor, a constant for each chemical reaction. According to collision theory, A is the frequency of collisions in the correct orientation,
- Ea is the activation energy for the reaction (in the same units as RT),
- R is the universal gas constant.[1][2][3]
Alternatively, the equation may be expressed as
where
- Ea is the activation energy for the reaction (in the same units as kBT),
- kB is the Boltzmann constant.
The only difference is the energy units of Ea: the former form uses energy per mole, which is common in chemistry, while the latter form uses energy per molecule directly, which is common in physics.The different units are accounted for in using either the gas constant, R, or the Boltzmann constant, kB, as the multiplier of temperature T.
The units of the pre-exponential factor A are identical to those of the rate constant and will vary depending on the order of the reaction. If the reaction is first order it has the units: s−1, and for that reason it is often called the frequency factor or attempt frequency of the reaction. Most simply, k is the number of collisions that result in a reaction per second, A is the number of collisions (leading to a reaction or not) per second occurring with the proper orientation to react[5] and is the probability that any given collision will result in a reaction. It can be seen that either increasing the temperature or decreasing the activation energy (for example through the use of catalysts) will result in an increase in rate of reaction.
Given the small temperature range of kinetic studies, it is reasonable to approximate the activation energy as being independent of the temperature. Similarly, under a wide range of practical conditions, the weak temperature dependence of the pre-exponential factor is negligible compared to the temperature dependence of the factor; except in the case of 'barrierless' diffusion-limited reactions, in which case the pre-exponential factor is dominant and is directly observable.
Arrhenius plot[edit]
Taking the natural logarithm of Arrhenius equation yields:
Rearranging yields:
This has the same form as an equation for a straight line:
where x is the reciprocal of T.
So, when a reaction has a rate constant that obeys Arrhenius equation, a plot of ln k versus T−1 gives a straight line, whose gradient and intercept can be used to determine Ea and A . This procedure has become so common in experimental chemical kinetics that practitioners have taken to using it to define the activation energy for a reaction. That is the activation energy is defined to be (−R) times the slope of a plot of ln k vs. (1/T):
Modified Arrhenius equation[edit]
The modified Arrhenius equation[6] makes explicit the temperature dependence of the pre-exponential factor. The modified equation is usually of the form
The original Arrhenius expression above corresponds to n = 0. Fitted rate constants typically lie in the range −1 < n < 1. Theoretical analyses yield various predictions for n. It has been pointed out that 'it is not feasible to establish, on the basis of temperature studies of the rate constant, whether the predicted T1/2 dependence of the pre-exponential factor is observed experimentally'.[4]:190 However, if additional evidence is available, from theory and/or from experiment (such as density dependence), there is no obstacle to incisive tests of the Arrhenius law.
Another common modification is the stretched exponential form[citation needed]
where β is a dimensionless number of order 1. This is typically regarded as a purely empirical correction or fudge factor to make the model fit the data, but can have theoretical meaning, for example showing the presence of a range of activation energies or in special cases like the Mott variable range hopping.
Theoretical interpretation of the equation[edit]
Arrhenius's concept of activation energy[edit]
Arrhenius argued that for reactants to transform into products, they must first acquire a minimum amount of energy, called the activation energy Ea. At an absolute temperature T, the fraction of molecules that have a kinetic energy greater than Ea can be calculated from statistical mechanics. The concept of activation energy explains the exponential nature of the relationship, and in one way or another, it is present in all kinetic theories.
The calculations for reaction rate constants involve an energy averaging over a Maxwell–Boltzmann distribution with as lower bound and so are often of the type of incomplete gamma functions, which turn out to be proportional to .
Collision theory[edit]
One approach is the collision theory of chemical reactions, developed by Max Trautz and William Lewis in the years 1916–18. In this theory, molecules are supposed to react if they collide with a relative kinetic energy along their line of centers that exceeds Ea. The number of binary collisions between two unlike molecules per second per unit volume is found to be[7]
where NA and NB are the number of molecules of A and B per unit volume, dAB is the average diameter of A and B, kB is the Boltzmann constant, and μ is the reduced mass.
The rate constant is then calculated as so that the collision theory predicts that the pre-exponential factor is equal to the collision number ZAB. However for many reactions this agrees poorly with experiment, so the rate constant is written instead as . Here P is an empirical steric factor, often much less than 1, which is interpreted as the fraction of sufficiently energetic collisions in which the two molecules have the correct mutual orientation to react.[7]
Transition state theory[edit]
The Eyring equation, another Arrhenius-like expression, appears in the 'transition state theory' of chemical reactions, formulated by Wigner, Eyring, Polanyi and Evans in the 1930s. The Eyring equation can be written:
Rearranging Arrhenius Equation To Find Activation Energy Without A Graph
where = the Gibbs energy of activation, is the entropy of activation, is the enthalpy of activation, is Boltzmann's constant, and is Planck's constant.[8]
At first sight this looks like an exponential multiplied by a factor that is linear in temperature. However, free energy is itself a temperature dependent quantity. The free energy of activation is the difference of an enthalpy term and an entropy term multiplied by the absolute temperature. The pre-exponential factor depends primarily on the entropy of activation. The overall expression again takes the form of an Arrhenius exponential (of enthalpy rather than energy) multiplied by a slowly varying function of T. The precise form of the temperature dependence depends upon the reaction, and can be calculated using formulas from statistical mechanics involving the partition functions of the reactants and of the activated complex.
Limitations of the idea of Arrhenius activation energy[edit]
Both the Arrhenius activation energy and the rate constant k are experimentally determined, and represent macroscopic reaction-specific parameters that are not simply related to threshold energies and the success of individual collisions at the molecular level. Consider a particular collision (an elementary reaction) between molecules A and B. The collision angle, the relative translational energy, the internal (particularly vibrational) energy will all determine the chance that the collision will produce a product molecule AB. Macroscopic measurements of E and k are the result of many individual collisions with differing collision parameters. To probe reaction rates at molecular level, experiments are conducted under near-collisional conditions and this subject is often called molecular reaction dynamics.[9]
There are deviations from the Arrhenius law during the glass transition in all classes of glass-forming matter.[10] The Arrhenius law predicts that the motion of the structural units (atoms, molecules, ions, etc.) should slow down at a slower rate through the glass transition than is experimentally observed. In other words, the structural units slow down at a faster rate than is predicted by the Arrhenius law. This observation is made reasonable assuming that the units must overcome an energy barrier by means of a thermal activation energy. The thermal energy must be high enough to allow for translational motion of the units which leads to viscous flow of the material.
See also[edit]
- Cherry blossom front – predicted using the Arrhenius equation
References[edit]
- ^ abArrhenius, S. A. (1889). 'Über die Dissociationswärme und den Einfluß der Temperatur auf den Dissociationsgrad der Elektrolyte'(PDF). Z. Phys. Chem.4: 96–116. doi:10.1515/zpch-1889-0408.
- ^ abArrhenius, S. A. (1889). 'Über die Reaktionsgeschwindigkeit bei der Inversion von Rohrzucker durch Säuren'. Z. Phys. Chem.4: 226–248. doi:10.1515/zpch-1889-0116.
- ^ abLaidler, K. J. (1987) Chemical Kinetics, Third Edition, Harper & Row, p.42
- ^ abKenneth Connors, Chemical Kinetics, 1990, VCH Publishers Chemical Kinetics: The Study of Reaction Rates in Solution at Google Books
- ^Silberberg, Martin S. (2006). Chemistry (fourth ed.). NY: McGraw-Hill. p. 696. ISBN0--07-111658-3.
- ^IUPAC Goldbook definition of modified Arrhenius equation.
- ^ abLaidler, Keith J.; Meiser, John H. (1982). Physical Chemistry (1st ed.). Benjamin/Cummings. pp. 376–8. ISBN0-8053-5682-7.
- ^Laidler, Keith J.; Meiser, John H. (1982). Physical Chemistry (1st ed.). Benjamin/Cummings. pp. 378–383. ISBN0-8053-5682-7.
- ^Levine, R.D. (2005) Molecular Reaction Dynamics, Cambridge University Press
- ^Bauer, Th.; Lunkenheimer, P.; Loidl, A. (2013). 'Cooperativity and the Freezing of Molecular Motion at the Glass Transition'. Physical Review Letters. 111 (22): 225702. arXiv:1306.4630. Bibcode:2013PhRvL.111v5702B. doi:10.1103/PhysRevLett.111.225702. PMID24329455.
Bibliography[edit]
- Pauling, L. C. (1988). General Chemistry. Dover Publications.
- Laidler, K. J. (1987). Chemical Kinetics (3rd ed.). Harper & Row.
- Laidler, K. J. (1993). The World of Physical Chemistry. Oxford University Press.
External links[edit]
- Carbon Dioxide solubility in Polyethylene – Using Arrhenius equation for calculating species solubility in polymers